Simulands, a company located in Zurich, has received €10 million in funding to enhance patient outcomes through simulation-based medical training.
The company will now accelerate the product’s commercialization.
Practice makes perfect, as they say. Repeated practice can help an activity be done better by fostering greater confidence and efficiency. It’s a typical method of teaching in many industries, and it’s currently being improved in the field of medicine.
Simulands, headquartered in Zurich, is working to reduce the risk of complex interventional procedures by improving how cardiologists and echocardiographers prepare for them.
According to the World Health Organization, cardiovascular disease is Europe’s main cause of premature death. Cardiologists perform cardiac surgery regularly, but there are few chances to practice the procedures; therefore, a more effective method of instruction is required. The Switzerland startup has just received €10 million from SHS to fulfill this need.
Medical device startup, QV Bioelectronics snaps £2M
The practice of medicine is being advanced by technological and scientific advancements, according to Andrea Guidotti, CEO of Simulands. We should reconsider how medical education is delivered, giving doctors new resources and methods for on-demand training and ongoing learning that are suited to their requirements and concentrated on the roles and competencies they must possess.
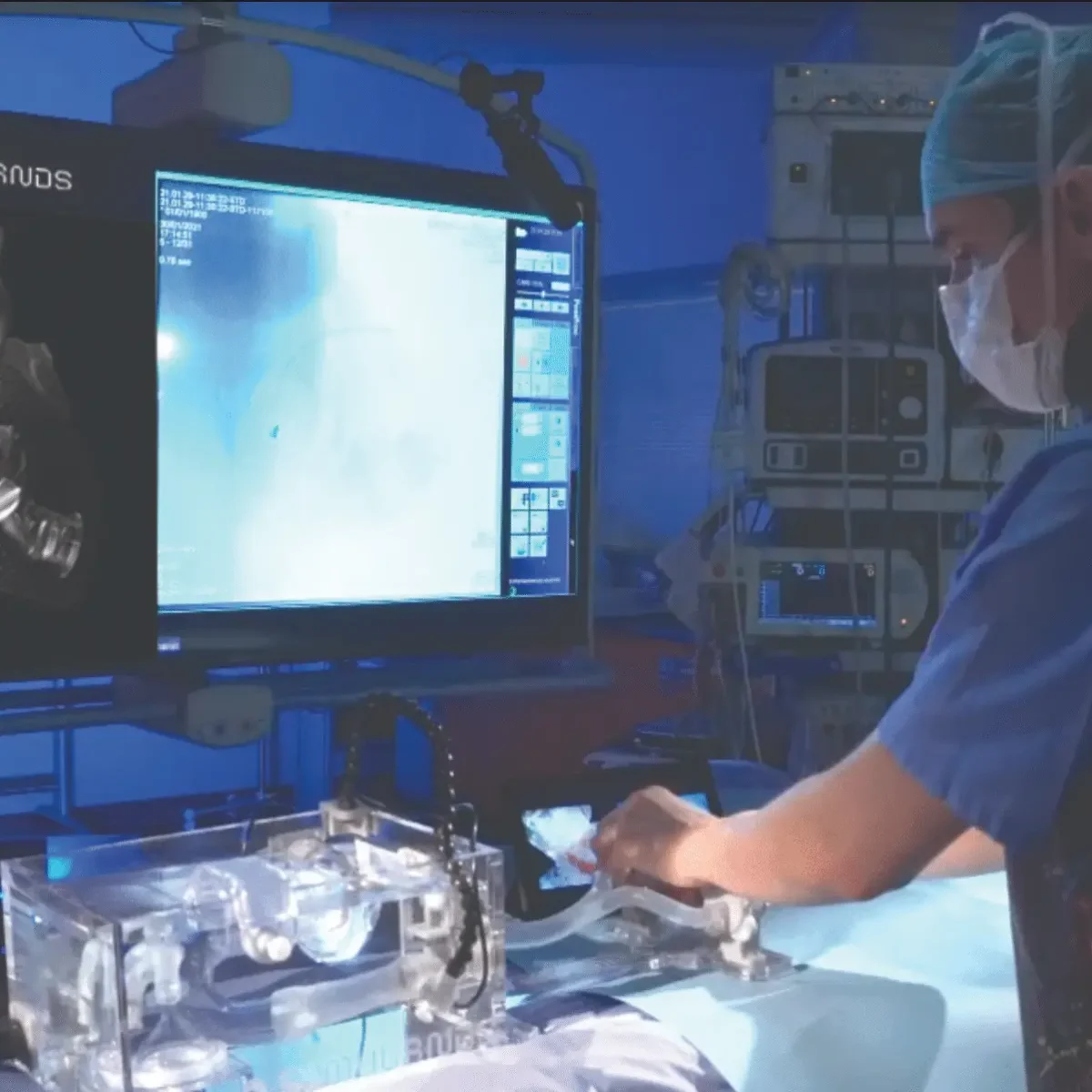
Simulands, a Zurich-based company, has created a medical-grade, ISO-compliant human-grade emulator. In the area of cardiovascular research, it can be used to reduce training and research conducted directly on humans and animals, making medical education more moral and procedures ultimately safer.
The startup’s solution gives procedure experience that caters to a broad range of experience and knowledge levels, providing accurate haptic responses, real procedural imaging guidance, and a true understanding of device performance. By establishing a secure learning atmosphere, the business provides operators with solutions for improving their skills and boosting doctors’ confidence and competence.
The simulators are built using additive manufacturing methods that accurately reproduce pathological anatomy and enable training with actual devices for cardiovascular minimally invasive treatments.
The company, founded in 2019, has been expanding quickly and has already built a solid reputation for its medical simulation training tools. Abbott Laboratories (NYSE: ABT), Edwards Lifesciences (NYSE: EW), and Boston Scientific are among its current customers (NYSE: BSX).
Andrea Guidotti: “Partnering with SHS is a priceless chance for Simulands to expand support to its customer base and accelerate entry into important international markets. SHS’ experience and track record of success in the healthcare industry will further solidify our development and hasten the realization of our long-term goals.
The company’s commercialization and the expansion of its mission to transform cardiovascular education and training will both benefit from this new funding.
SHS partner Dr. Cornelius Maas: “We are happy to make our first investment from our recently established sixth fund generation with the investment into Simulands AG. We feel strongly that the business should be supported in its efforts to improve cardiological interventions for both physicians and patients.”
He added, “With our investment, we particularly want to promote both international and product portfolio expansion. We are looking for additional cutting-edge, rapidly expanding healthcare businesses that can significantly improve healthcare for our new fund.”